
Placebo plus cisplatin (80 mg/m 2 IV on Day 1 of each three-week cycle for up to six cycles) plus 5-FU (800 mg/m 2 IV per day on Day 1 to Day 5 of each three-week cycle, or per local standard for 5-FU administration, for up to 35 cycles).Įsophageal cancer, a type of cancer that is particularly difficult to treat, begins in the inner layer (mucosa) of the esophagus and grows outward. KEYTRUDA (200 mg intravenously on Day 1 of each three-week cycle for up to 35 cycles) plus cisplatin (80 mg/m 2 IV on Day 1 of each three-week cycle for up to six cycles) plus 5-FU (800 mg/m 2 IV per day on Day 1 to Day 5 of each three-week cycle, or per local standard for 5-FU administration, for up to 35 cycles) or The study enrolled 749 patients who were randomized to receive either: 
Additional efficacy outcome measures were objective response rate and duration of response. The major efficacy outcome measures were OS and PFS. KEYNOTE-590 is a Phase 3, randomized, double-blind trial (, NCT03189719) evaluating KEYTRUDA in combination with platinum and fluoropyrimidine based chemotherapy versus placebo plus chemotherapy (cisplatin plus 5-fluorouracil ) for the first-line treatment of patients with locally advanced unresectable or metastatic carcinoma of the esophagus and GEJ. Merck is continuing to study KEYTRUDA across multiple settings and stages of gastrointestinal cancer – including gastric, hepatobiliary, esophageal, pancreatic, colorectal and anal cancers – through its broad clinical program. KEYTRUDA is currently approved in the U.S., China and Japan as monotherapy for the second-line treatment of patients with recurrent locally advanced or metastatic squamous cell carcinoma of the esophagus whose tumors express PD-L1 (Combined Positive Score ≥10). “We look forward to working with the FDA to bring a new option to patients in the first-line setting.” Vicki Goodman, vice president, clinical research, Merck Research Laboratories. “Patients with newly diagnosed esophageal and GEJ cancer face an aggressive disease with a poor prognosis, despite the currently available treatment options,” said Dr. The FDA has set a Prescription Drug User Fee Act (PDUFA), or target action, date of April 13, 2021. These data were presented at the European Society for Medical Oncology (ESMO) Virtual Congress 2020. 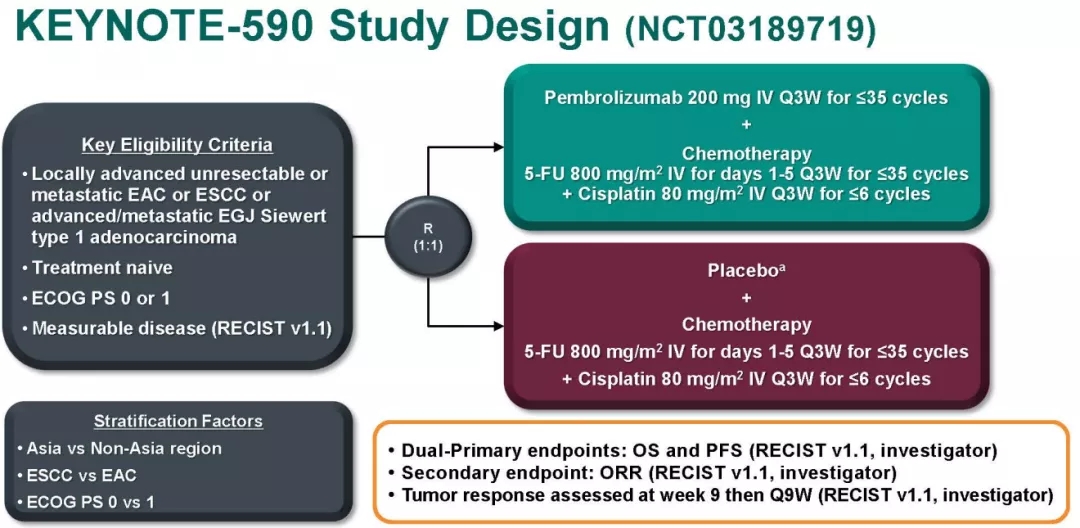
This sBLA is based on data from the pivotal Phase 3 KEYNOTE-590 trial, in which KEYTRUDA plus chemotherapy demonstrated significant improvements in the primary endpoints – overall survival (OS) and progression-free survival (PFS) – versus chemotherapy in these patients regardless of PD-L1 expression status and tumor histology. 
Food and Drug Administration (FDA) has accepted and granted priority review for a new supplemental Biologics License Application (sBLA) for KEYTRUDA, Merck’s anti-PD-1 therapy, in combination with platinum and fluoropyrimidine based chemotherapy for the first-line treatment of patients with locally advanced unresectable or metastatic carcinoma of the esophagus and gastroesophageal junction (GEJ). Merck (NYSE: MRK), known as MSD outside the United States and Canada, today announced that the U.S. Application Based on Overall Survival and Progression-Free Survival Data Comparing KEYTRUDA Plus Chemotherapy to Chemotherapy Alone From Pivotal Phase 3 KEYNOTE-590 Trial
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
